Corrosion-resistant alloys rely upon the presence of a passive chromium oxide film on the surface to provide corrosion protection. The presence of chlorides or oxygen may result in this film becoming unstable. Small areas will then become active resulting in rapid localized corrosion creating small holes in the metal. These holes are difficult to detect and may lead to catastrophic failure. Elevated temperature and/or low pH will also reduce the stability of the chromium oxide film and may therefore be contributing factors.
Resistance to pitting corrosion is achieved by increasing the chromium content or alloying with molybdenum, tungsten or nitrogen. A common tool for ranking the pitting resistance of CRAs is the use of an empirical formula to calculate the Pitting Resistance Equivalent Number (PREN). The most common form of this equation is:
PREN = CRwt% + 3.3 x (Mowt% + 0.5 x Wwt%) + 16 x Nwt%
Presented below is a comparison of PREN values for corrosion-resistant alloys commonly used for oil & gas well completions.
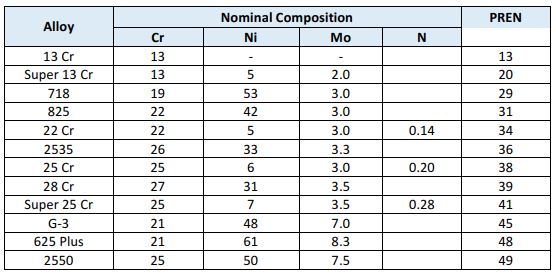

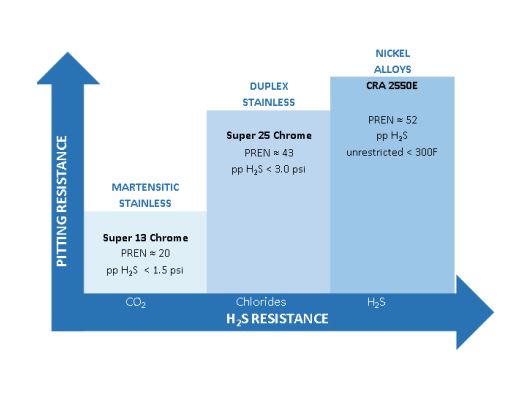
If the chloride content exceeds 100,000 ppm or oxygen content exceeds 10 ppb, corrosion-resistant alloys with a high Pitting Resistance Equivalent Number (PREN) should be used. The martensitic stainless steels will not provide sufficient protection. To prevent localized corrosion in seawater or where there is a potential for oxygen contamination, an alloy with PREN>40 is recommended, such as Super 25Cr or CRA 2550E.
Corrosion pits in the presence of tensile stresses may act as stress risers and serve as initiation sites for Stress Corrosion Cracking (SCC). Alloys with higher nickel content are more resistant to SCC. Alloys with nickel content greater than 42%, such as CRA 2550E are considered immune to SCC from chlorides.

*While every effort has been made to ensure the accuracy of the above review, assessment, conclusions, and report, the appropriateness of their application and their interpretation remain the sole responsibility of the user.
"*" indicates required fields